|
But what I got is about 0.2 or so.Ĭould anyone help me fix it right? I think the problem should be in the formula of variable cp. 
The value of Cp should be 4.184kJ/kgK for water at 300K&1atm. Variable cp equal (f_3-f_2^2)/NA/kB/T/T/18 # kJ/kgK (I removed dollar sign for simplicity) Variable h equal abs(enthalpy)*4184/900 # J/mol Here is my related LAMMPS commands, (my system has 900 water molecules) It is essentially a value for the specific enthalpy. In kilograms, this amounts to 2257 kJ/kg. I guess the main reason is the unit conversion process during calculation. An online enthalpy calculator is specially designed to calculate exact amount of enthalpy generated in a thermodynamic system. From experimental data, this number is known to be 40.65 kJ/mol. However, I just cannot get the right results. 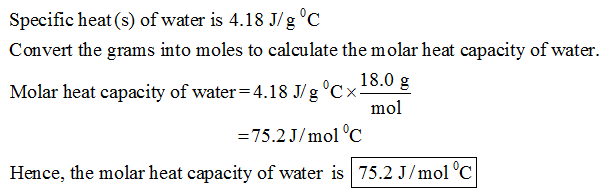 
Based on the above equation, I wrote my own LAMMPS commands to calculate Cp of water at 300K&1atm.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
